The patent US12097187B2 is currently at the heart of litigation filed by Allergan Holdings Unlimited Company and others, targeting MSN Laboratories and Hetero USA. While Allergan operates as a non-practicing entity, the dispute involves two specific drug products alleged to infringe on this single patent-in-suit.
At its core, this patent covers oral dosage formulations of an opioid receptor modulator specifically designed to treat Irritable Bowel Syndrome with Diarrhea (IBS-D). Importantly, the patent emphasizes abuse-deterrent properties, ensuring the drug remains effective without being easily tampered with or misused, an essential safeguard in opioid-related therapies.
This patent is being targeted because it potentially blocks generic competition. If the formulation is protected, others can’t bring similar abuse-deterrent IBS treatments to market without risking infringement, making it a critical point of contention in the pharmaceutical space.
To assess its strength, finding related patents is essential, and that’s where the Global Patent Search (GPS) tool comes in. By entering the patent number, GPS uncovers similar technologies, maps key features, and delivers structured comparisons that help you evaluate how this patent holds up in a competitive landscape.
Understanding Patent US12097187B2
Patent US12097187B2 focuses on oral abuse-deterrent formulations containing a specific opioid receptor modulator: 5-({[(2S)-2-Amino-3-(4-carbamoyl-2,6-dimethyl-phenyl)-propionyl]-[(1S)-1-(4-phenyl-1H-imidazol-2-yl)-ethyl]-amino}-methyl)-2-methoxy-benzoic acid).
This compound is designed to treat Irritable Bowel Syndrome with Diarrhea (IBS-D) by simultaneously activating mu-opioid receptors (MOR) and blocking delta-opioid receptors (DOR). It delivers pain relief and improved bowel function without the constipating side effects of traditional opioids. The patent emphasizes formulations resistant to misuse or abuse, a growing concern with opioid-class treatments.
Its four key features are:
#1. Dual-acting opioid modulation: Agonist of MOR and antagonist of DOR for balanced gastrointestinal control.
#2. Oral formulation with abuse deterrence: Structured to resist crushing, tampering, or chemical manipulation.
#3. Targeted for IBS-D: Designed to reduce abdominal pain and normalize stool consistency.
#4. Fixed dosage method: 75 mg administered twice daily with food, showing clinical improvement in pain and bowel symptoms.
This patent is central to the litigation because it protects a specific, abuse-resistant formulation of a compound used for IBS-D, a therapeutic area with limited, safe options. By targeting this patent, defendants may attempt to invalidate exclusivity rights that block competing generic or alternative formulations from entering the market. With its formulation-based claims, the patent is a potential barrier to entry, especially for companies developing similar gastrointestinal treatments.
Related Patent References for US12097187B2
#1. CA2359273A1
CA2359273A1, filed on January 17, 2000, by Grünenthal GmbH, describes pharmaceutical formulations containing an opioid and an α-agonist. The invention aims to reduce side effects associated with opioid therapy by combining active agents that balance efficacy with tolerability. These formulations treat moderate to severe pain through oral, parenteral, or transdermal administration.
Key Features of this Related Patent:
- Opioid and α-agonist combination – The invention uses an opioid paired with an α-agonist to reduce adverse effects while maintaining analgesic action.
- Formulations suitable for oral administration – Includes immediate and controlled-release profiles to optimize therapeutic response.
- Focus on abuse resistance and tolerability – Intended to offer effective pain relief with a lower risk of misuse and opioid-related side effects.
- Therapeutic use in managing moderate to severe pain – Targeted at chronic pain patients with higher treatment needs.
Which features of US12097187B2 are disclosed by CA2359273A1?
| Key Feature of Claim | Disclosure Status |
| The administration frequency is twice daily | Partially Disclosed |
Key Excerpt from CA2359273A1:
“The pharmaceutical formulation according to the invention may also contain at least one of the active substances in controlled release form as well as in non-controlled release form… Combination with the immediately released active substance allows therapeutic coverage that may extend to twice daily administration.”
#2. ES2278647T3
ES2278647T3, filed on November 29, 2000, by Adolor Corp, discloses new pharmaceutical procedures and compositions that include combinations of opioids and opioid antagonists. The invention focuses on minimizing gastrointestinal side effects of opioid treatments, such as constipation and nausea, by using peripherally selective antagonists alongside opioid compounds. The goal is to improve tolerability without compromising analgesic effectiveness.
Key Features of this Related Patent:
- Opioid-antagonist combinations – Designed to maintain opioid efficacy while reducing peripheral side effects.
- Treatment for opioid-induced gastrointestinal symptoms – Including constipation, nausea, and motility issues.
- Oral dosage forms – Tablets or capsules combining active agents, potentially allowing controlled delivery.
- Clinical use cases evaluated – Phase I studies explored human GI transit impact and formulation performance.
Which features of US12097187B2 are disclosed by ES2278647T3?
| Key Feature of Claim | Disclosure Status |
| The administration frequency is twice daily | Partially Disclosed |
Key Excerpt from ES2278647T3:
“As for the typical dosage form of this type of combination product, such as a tablet, the opioid compounds (for example, morphine) are generally present in an amount of 15 to 200 milligrams… A Phase I study with 11 volunteers demonstrated that an oral dose of 3 mg of the compound of formula (II), administered three times a day, blocked the inhibition of gastrointestinal transit produced by oral delayed-release morphine.”
#3. JP2003534380A
JP2003534380A, filed on May 10, 2001, by Grünenthal GmbH, discloses a combination of active substances comprising at least one opioid compound and a second component based on a general formula I. This invention is intended to treat various pain conditions and aims to improve therapeutic outcomes through multi-target drug combinations. The compositions are suitable for multiple delivery routes and provide flexibility in dosage forms and patient-specific treatments.
Key Features of this Related Patent:
- Combination drug formulations – Includes opioids and structurally related compounds based on a general formula.
- Wide dosage range – From 0.1 mg to 2000 mg, adaptable to various administration methods.
- Potential twice-daily administration – Dosage scheduling may include up to twice-daily release.
- Multi-route delivery – Including oral, intravenous, subcutaneous, and others.
Which features of US12097187B2 are disclosed by JP2003534380A?
| Key Feature of Claim | Disclosure Status |
| The method comprises oral administration of 5-({[(2S)-2-Amino-3-(4-carbamoyl-2,6-dimethyl-phenyl)-propionyl]-[(1S)-1-(4-phenyl-1H-imidazole-2-yl)-ethyl]-amino}-methyl)-2-methoxy-benzoic acid | Partially Disclosed |
| The administration frequency is twice daily | Partially Disclosed |
Key Excerpt from JP2003534380A:
“In the case of once-daily administration, the combination of active substances of the present invention preferably contains 0.1 to 2000 mg… The release of the dose to be administered can be carried out once or up to twice daily, depending on formulation and patient need.”
#4. JPH1112166A
JPH1112166A, filed on March 24, 1998, by Grünenthal GmbH, discloses the oral administration of (+)-O-demethyltramadol as an analgesic for treating acute and chronic pain. The invention centers on optimizing the delivery and therapeutic response of this opioid analgesic when taken orally, offering potential alternatives to conventional tramadol formulations.
Key Features of this Related Patent:
- Opioid-based analgesic – Focused on (+)-O-demethyltramadol, a potent opioid derivative.
- Oral administration route – Designed specifically for oral delivery to improve patient compliance.
- Variable dosing strategy – Doses ranging from 10–400 mg/kg depending on severity and patient profile.
- Therapeutic scope covers general pain management – No specific indication for gastrointestinal disorders.
This is how feature mapping from the tool looks like:
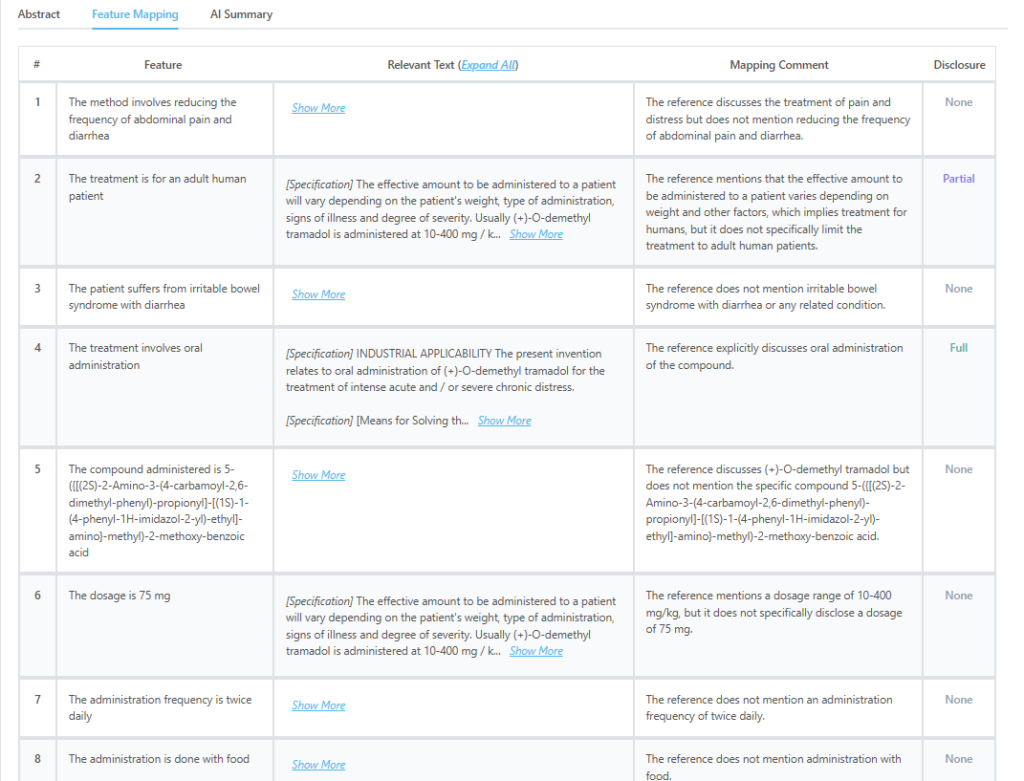
Source: GPS
Which features of US12097187B2 are disclosed by JPH1112166A?
| Key Feature of Claim | Disclosure Status |
| The treatment is for an adult human patient | Partially Disclosed |
| The treatment involves oral administration | Fully Disclosed |
Key Excerpt from JPH1112166A:
“The present invention relates to oral administration of (+)-O-demethyl tramadol for the treatment of intense acute and/or severe chronic distress… The effective amount to be administered to a patient will vary depending on the patient’s weight, type of administration, signs of illness and degree of severity.”
#5. TW476755B
TW476755B, filed on February 11, 1999, by Ortho-McNeil Pharmaceutical Inc, discloses a class of 4-[aryl(hexahydropyridin-4-yl)]aminobenzamides as δ-opioid receptor agonists/antagonists. The compounds are designed to treat various conditions, including diarrhea, gastritis, inflammation, and pain. Their therapeutic use includes potential application in central nervous system disorders and gastrointestinal conditions.
Key Features of this Related Patent:
- δ-opioid receptor agents – The compounds act as agonists or antagonists targeting delta-opioid receptors.
- Oral pharmaceutical formulations – The invention supports oral administration for effective therapeutic action.
- Multi-functional treatment applications – Covers gastrointestinal symptoms like diarrhea, along with pain management and CNS conditions.
- Flexible dosing – Describes potential dosing between 1-3000 mg and multiple daily frequencies.
Which features of US12097187B2 are disclosed by TW476755B?
| Key Feature of Claim | Disclosure Status |
| The method involves reducing the frequency of abdominal pain and diarrhea | Partially Disclosed |
| The treatment is for an adult human patient | Partially Disclosed |
| The treatment involves oral administration | Fully Disclosed |
| The administration frequency is twice daily | Partially Disclosed |
Key Excerpt from TW476755B:
“Another object of the present invention is to provide δ-opioid receptor agonists/antagonists as immunosuppressive agents, anti-inflammatory agents, agents for treating mental illness… as well as agents for treating gastrointestinal disorders such as diarrhea and gastritis.”
Feature Comparison Table
| Key Feature of Claim 1 | CA2359273A1 | ES2278647T3 | JP2003534380A | JPH1112166A | TW476755B |
| The method reduces the frequency of diarrhea in adult human patients with irritable bowel syndrome with diarrhea | Not Disclosed | Not Disclosed | Not Disclosed | Not Disclosed | Partially Disclosed |
| The method reduces the frequency of diarrhea in adult human patients with irritable bowel syndrome with diarrhea. | Not Disclosed | Not Disclosed | Not Disclosed | Not Disclosed | Partially Disclosed |
| The method reduces the frequency of diarrhea in adult human patients with irritable bowel syndrome with diarrhea. | Not Disclosed | Not Disclosed | Partially Disclosed | Not Disclosed | Not Disclosed |
| The dosage is 75 mg. | Not Disclosed | Not Disclosed | Not Disclosed | Not Disclosed | Not Disclosed |
| The administration frequency is twice daily. | Partially Disclosed | Partially Disclosed | Partially Disclosed | Not Disclosed | Partially Disclosed |
| The drug is administered with food. | Not Disclosed | Not Disclosed | Not Disclosed | Not Disclosed | Not Disclosed |
How to Find Related Patents Using Global Patent Search
Uncovering related patents is critical when evaluating the strength, novelty, or potential vulnerability of a patent like US12097187B2. The Global Patent Search tool simplifies this process with a fast, structured, and data-driven approach. Here’s how GPS helps:
Search by patent number or description – Quickly locate technologies with similar functions, formulations, or therapeutic goals.
Source: GPS
Feature-level mapping – Compare specific claim elements from the patent-in-suit to existing disclosures across global filings.
Detailed matching reports – Instantly see which features are disclosed, partially disclosed, or missing in each related reference.
Confidence in strategy – Use GPS insights to shape litigation arguments, assess patent validity, or guide competitive R&D.
Whether preparing for a challenge or defending a portfolio, GPS provides the clarity you need faster and more accurately than manual searching.
Take the Guesswork Out of Related Patent Research
Patent litigation is complex, but identifying related patents doesn’t have to be. The Global Patent Search tool empowers legal teams, researchers, and innovators with:
- Instant results – No more manual sifting through thousands of documents.
- Accurate feature mapping – See exactly how each patent compares, line by line.
- Data-driven insights – Make informed decisions with clear, structured results.
If you are assessing a patent’s strength, defending a formulation, or challenging exclusivity, GPS gives you the edge. Start your search with Global Patent Search today because every detail matters.
Disclaimer: The information provided in this article is for informational purposes only and should not be considered legal advice. The related patent references mentioned are preliminary results from the Global Patent Search tool and do not guarantee legal significance. For a comprehensive related patent analysis, we recommend conducting a detailed search using GPS or consulting a patent attorney.




